
Home List your patent My account Help Support us
Colostomy closure device, prototypes available
[Category : - HEALTH]
[Viewed 653 times]
Financial information
StomAid®)
The project requires financial partners willing to finance the completion of the project until the achievement of the following listed objectives step by step
Assuming StomAid®) potentially substitute stoma bags market:
Italian ANNUAL STOMA BAGS DEMAND is:
Est. living colostomates n° 21,500
Est. Stoma Bags monthly consumption/pers n° 30 to 60
Est. Stoma Bags annual consumption/pers n° 360 to 720
ANNUAL STOMA BAGS SALES
Est. Stoma Bags annual expenditure /pers - € 1,080 to 2,160
Est. Stoma Bags business volume/year - € 23 to 46 millions
REFINANCING THE COMPANY
Budget (rough)
a) Skills ~ € 276,000.00
b) Device
i) Body no. 100 items ~ € 00
ii) Balloon, n. 100 items, ~ € 17,500.00
iii) Filters (New model)n. 100 items ~ € 17,500.00
iv) Assembly, packaging, sterilization n. 100 items ~ € n.a.
c) CE ~ € 200,000.00
______________________________________________________________ Budget Sub TOTAL ~ € 511,100.00
Sundry payables ~ € 1,428,000.00
______________________________________________________________
ENTRY-FEE (i.e.proprietary capital employed) ~ € 1,939,100.00
______________________________________________________________
MEDICEA MEDICA SRL-
To date, the activity carried out by the company is that of research in the field of biotechnology as it is developing a prototype for enterostomized patients with the Stomaid brand and it is for this reason that the company is still inactive; once the prototype has been defined and the experimentation completed and the CE certification has been obtained, the company will be able to start the production and sale of the device.
-The production will be completely outsourced while the management of order collection and sales will be managed directly by the company.
-The prototype in the definition phase derives from the granting of the license to the company of a patent owned by Dr. Bencini.
Considered to have a purely technological and innovative vocation, the company's assets consist exclusively of intangible fixed assets listed below:
-Start-up and expansion costs:
*these are the costs of setting up the company.
-Development costs:
*made up of the costs incurred for the development of the prototype. -Patent License:
*This is the registered patent relating to the medical device, which
value is quantified on the basis of the licensing agreement.
-Trademark:
*these are expenses incurred for the registration of the StomAid trademark.
-The aforementioned intangible fixed assets, as required by the national accounting standards, are valued at cost, but it is easy to understand that, once the production and distribution of the StomAid medical device has begun, seen at its revolutionary extent in terms of the quality of life of the enterostomized subjects, both the patent and the trademark will have a much higher market value considering
above all the fact that this patent has been registered at an international level.
-The liabilities, on the other hand, consist of:
*Sundry payables, whose amount of € 1,428,000.00 is equal to the book value of the patent, as regards the amount that the company will have to pay to Dr. Bencini, owner-licensee of the same starting from the marketing of the StomAid®) device; Payables to shareholder Bencini, as Dr. Bencini financed the costs inherent in the experimental phase.
-The additional costs involved in bringing the product to market consist of € 511,100.00
THEREFORE ENTRY-FEE is ~ € 1,939,100.00
Asking price:
Above 1 million USD
Above 1 million USD


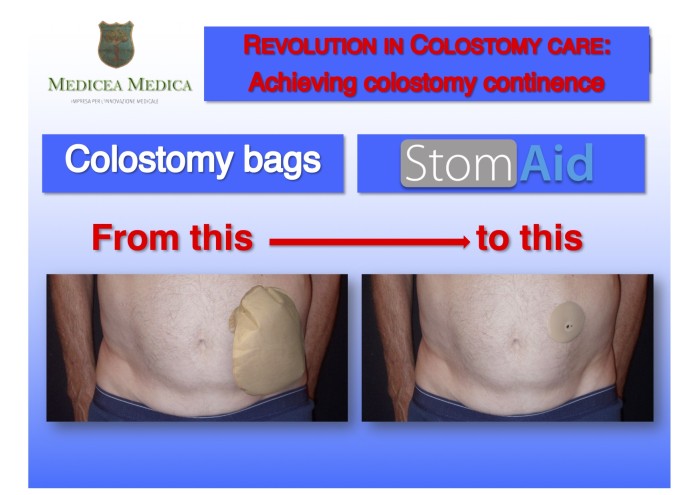
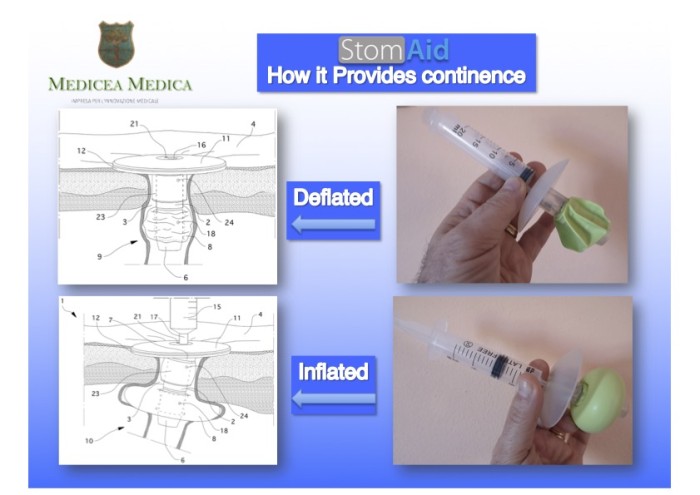
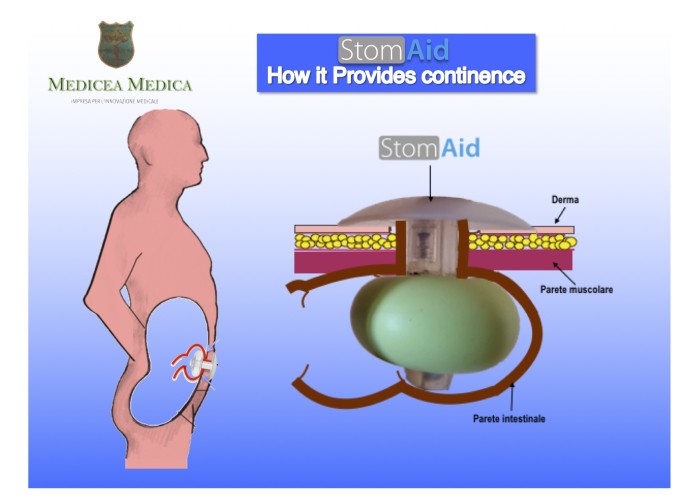
[ Home | List a patent | Manage your account | F.A.Q.|Terms of use | Contact us]
Copyright PatentAuction.com 2004-2017
Page created at 2025-12-19 7:13:33, Patent Auction Time.
 Patent publications:
Patent publications: US 20180353319
US 20180353319 Great invention
Great invention